
UCLA researchers receive $13.9 million CIRM grant to identify drug targets for autism and schizophrenia using human stem cell models
A team of researchers led by UCLA’s Dr. Daniel Geschwind has received a $13.9 million grant from the California Institute for Regenerative Medicine to use human stem cell-based models to uncover the molecular and cellular mechanisms underlying autism and schizophrenia and to identify drug targets for both conditions.
These conditions affect tens of millions of people in the U.S., yet no drugs exist that target their underlying biology — largely because researchers don't yet know precisely how brain cells from people with these conditions differ, at the molecular level, from those of neurotypical individuals, leaving nothing concrete to screen drugs against.
“Right now, there's virtually no roadmap for developing treatments for these conditions,” said Geschwind, a distinguished professor of human genetics, neurology and psychiatry at the David Geffen School of Medicine at UCLA and member of the UCLA Broad Stem Cell Research Center. “This work is focused on solving that problem by studying many variants in parallel, which will provide an initial roadmap for our lab, for other researchers, and for the pharmaceutical companies that could eventually take these findings into the clinic.”
Both conditions are thought to have strong genetic components that emerge as early as prenatal brain development, but those early molecular differences have never been systematically characterized across a representative patient population — leaving the drug development pipeline effectively stalled before it starts.
To address this gap, Geschwind and his multidisciplinary team will pursue two parallel scientific strategies, each targeting a different dimension of genetic risk. The first focuses on specific, well-documented genetic variants already known to raise the risk of autism or schizophrenia.
Using CRISPR gene editing, the team will introduce 36 autism-linked variants and 32 schizophrenia-linked variants — several of which are shared between the two conditions — into human stem cells, then coax those cells to grow into miniature, brain-like structures.
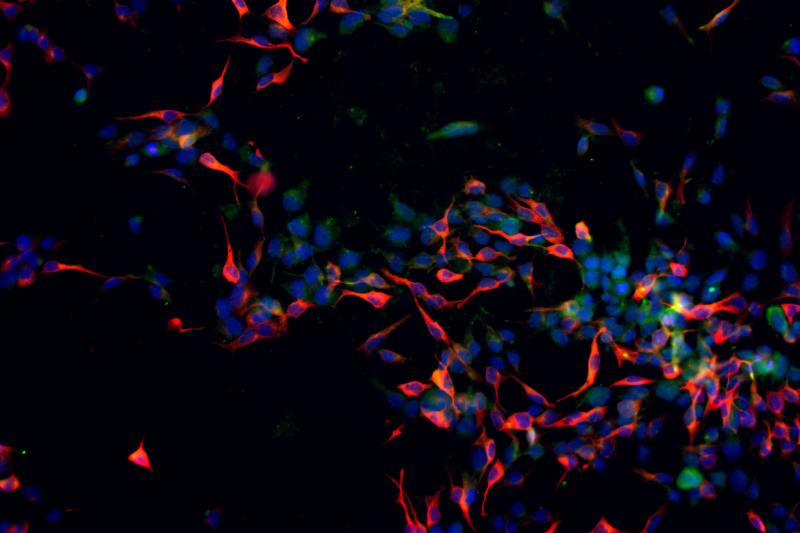
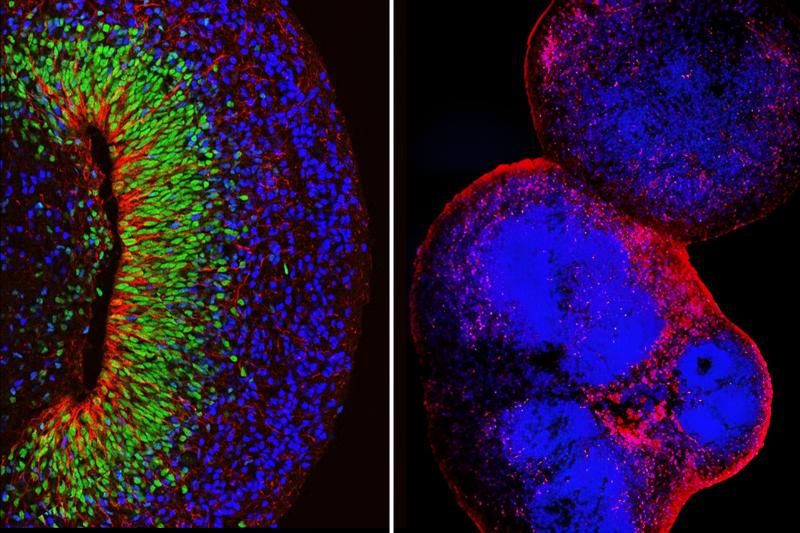
These structures come in two forms: organoids, tiny 3D tissue clusters that replicate the composition and architecture of parts of the developing brain, and assembloids, which are built by fusing multiple organoids together to model how distinct brain regions communicate. By studying the molecular, structural and electrical properties of these models, the team aims to pinpoint exactly how each genetic variant alters brain development.
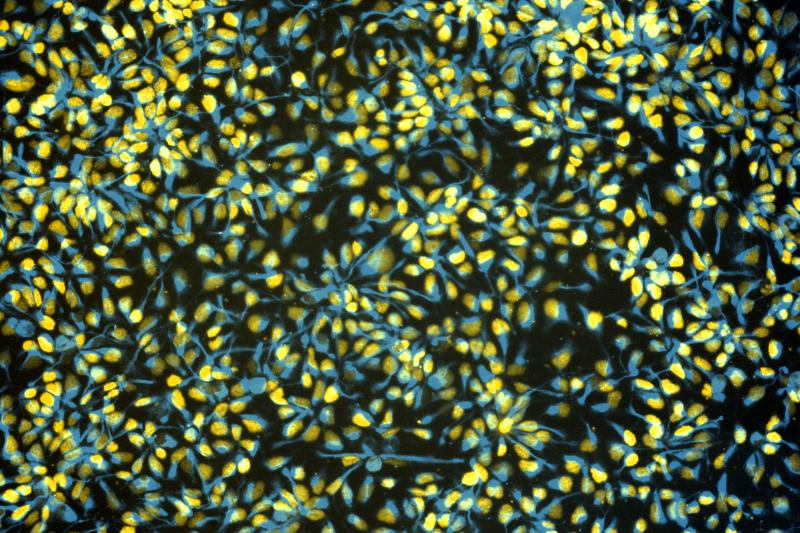
The second strategy addresses a harder and more common problem. In the vast majority of autism and schizophrenia cases, no single genetic variant can be identified as the cause. Studying cases where no single variant is responsible has historically been nearly impossible to do at meaningful scale in the lab.
Cell villages, a platform pioneered by Wells, are designed to solve exactly that problem. The researchers will use this approach to grow and examine stem cell-derived brain cells from more than 100 people with autism, more than 100 people with schizophrenia and more than 100 people without either condition together in a single shared dish.
When cells are cultured in separate wells, subtle but important biological differences can be drowned out by technical noise: variations in temperature, oxygen levels and evaporation that differ from well to well on the same plate. Growing all the samples together eliminates that noise entirely. And, because each person’s DNA is unique, it acts as a natural bar code, allowing researchers to identify which cell came from which donor and trace any biological differences back to their genetic source, even in a mixed population of hundreds.
“By putting everyone’s cells together in the same dish, we improve our ability to detect meaningful biological differences between patients and neurotypical controls,” said Dr. Michael F. Wells, a co-investigator on the grant and an assistant professor of human genetics at the David Geffen School of Medicine at UCLA. “We also reduce costs dramatically: what might otherwise require tens of thousands of sequencing samples can be done with dozens to hundreds.”
The two research tracks are designed to converge. Findings from the CRISPR-edited rare variant models will be compared against the cell village data to determine if genetically diverse forms of the two conditions share common underlying mechanisms — and whether those points of convergence could serve as viable drug targets. To validate those findings in a more structurally complex model, the team will also build assembloids from a representative selection of patient lines and compare them directly with the rare variant models from the first track.
Undergirding both tracks is a commitment to ensuring the findings from this work can benefit all patients with these conditions, regardless of their background. Studies to date have primarily relied on cell lines derived from individuals of European ancestry. In contrast, the studies supported by this grant will include cell lines from donors of Hispanic/Latino, African American and European backgrounds.
“For us it's all about representation,” Wells said. “Many drugs that work in one ancestral population do not work as well in others because those individuals were not included in the preclinical studies. At least here, we can ensure a broad array of genetic variation is included from the start.”
The project brings together a team of stem cell biologists, clinicians, bioinformaticians, electrophysiologists and data integration experts. Geschwind will provide clinical expertise as a world-renowned autism genetics expert; Wells will lead the cell village experiments; Dr. Robert Damoiseaux will lead automation and drug target translation; Dr. Bennett Novitch will drive the assembloid model experiments; Dr. Peyman Golshani and Dr. Mohammed Mostajo-Radji of UC Santa Cruz will lead electrophysiology studies in 2D cultures; Dr. Harold Pimentel will lead bioinformatic analysis.
If successful, the work could open the door to something that doesn't yet exist: a reliable way to screen drug compounds against autism and schizophrenia in the lab. In the final phase of the project, the team will test up to ten compounds in assembloid models of these conditions to see which, if any, can reverse the cellular differences observed. Those that do would represent strong candidates for further clinical investigation.
“It’s difficult to develop interventions without understanding the origins of these conditions,” Wells said. "By studying such a broad range of genetic contributors, our goal is to identify mechanisms and drug targets that are relevant to a much larger proportion of these patient populations — potentially enabling drug screens in a dish that could ultimately feed a clinical trial pipeline."



